
Physical properties of some common organic solvents
| α | β | ε | μ | Y | Z | ET | |
| Acetic Acid | 1.12 | 6.2 | 1.74 | -1.639 | 79.2 | 51.7 | |
| Water | 1.17 | 0.14 | 84.2 | 2.2 | 3.49 | 94.6 | 63.1 |
| Formamide | 110 | 3.7 | 0.6 | 83.2 | 56.6 | ||
| Methanol | 0.93 | 0.62 | 38.6 | 3.46 | -1.09 | 83.6 | 55.5 |
| Ethanol/water(80%) | 36 | 0 | 84.8 | 53.6 | |||
| Ethanol | 0.83 | 0.77 | 24.3 | 1.69 | -2.03 | 79.6 | 51.9 |
| Isopropyl alcohol | 0.76 | 0.92 | 18.3 | 1.66 | -2.73 | 76.3 | 48.6 |
| Acetonitrile | 36.2 | 3.92 | 71.3 | 46 | |||
| Dimethylsulfoxide | 0.743 | 49 | 3.96 | 71.1 | 45 | ||
| Sulfolane | 44 | 4.7 | 77.5 | 44 | |||
| t-Butanol | 0.68 | 0.95 | 10.9 | 1.66 | -3.26 | 71.3 | 43.9 |
| Dimethylformamide | 0.707 | 36.7 | 3.86 | 68.5 | 43.8 | ||
| Acetone | 0.507 | 20.7 | 2.88 | 65.7 | 42.2 | ||
| Dichloromethane | 8.9 | 1.6 | 64.2 | 41.1 | |||
| Pyridine | 0.668 | 12.3 | 2.19 | 64 | 40.2 | ||
| Chloroform | 4.7 | 1.87 | 63.2 | 39.1 | |||
| Hexamethylphosphoramide | 1.00 | 30 | 5.54 | 62.8 | 40.9 | ||
| Tetrahydrofuran | 0.553 | 7.32 | 1.63 | 37.4 | |||
| Diethyl ether | 0.514 | 4.34 | 1.15 | 34.6 | |||
| Benzene | 2.28 | 0 | 54 | 34.5 | |||
| Toluene | 2.38 | 0.36 | 33.9 | ||||
| Carbon tetrachloride | 2.23 | 0 | 32.5 | ||||
| Hexane | 1.89 | 0.08 | 30.9 |
Definitions:
α = describes the ability of a solvent to donate a proton in a solvent-to-solute hydrogen bond. Ref: Murray & Politzer, J. Org., Chem., 1991, 56, 6715-6717.
β = describes the ability of a solvent to accept a proton in a solute-to-solvent hydrogen bond. Ref: Taft et al., J. Am. Chem. Soc., 1976, 98, 377-383.
ε = dielectric constant, a macroscopic property of the solvent as a continuous medium. Ref: Wikipedia.
μ = dipole moment, debyes, a microscopic property of the individual molecule. Ref: Wikipedia.
Y = log(k(test solvent)/k(80%EtOH/water)) for SN1 reaction of tert-butyl chloride at 25°C. A more polar solvent makes this reaction faster, and Y more positive. Ref: J. Am. Chem. Soc., 1948, 70, 846-854.
Z = energy of the photochemical excitation of methyl N-ethyl-4-pyridinecarboxylate iodide, kcal/mol, in the particular solvent. Ref: E. M. Kosower, An Introduction to Physical Organic Chemistry (Wiley, New York, 1968).

ET = energy of the photochemical excitation of an N-phenoxypryidinium zwitterion, kcal/mol, in the particular solvent. Ref: Reichardt, Chem. Rev., 1994, 94, 2319-2358.
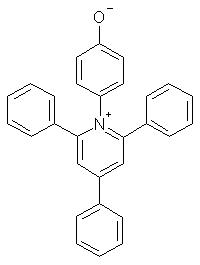
For both Z and ET, the stabilization of the polar reactant vs. the nonpolar excited state leads
to an increase in excitation energy in polar solvents.
Back to CH 362 Home page
02/28/05 ;