A. Calculate the resonance energy of an enolate anion using the following assumptions:
1. The Coulomb integral for oxygen is a+0.2b. This
reflects the greater electronegativity of oxygen versus carbon.
2. The resonance integral for a C-O interaction is 0.8b,
reflecting the fact that a C=O p bond is weaker than a C=C p bond.
Hint: set up the secular determinant using explicit definitions, rather than the conventional substitution of x = (a-E)/b.
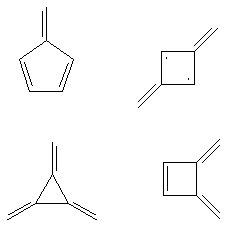
B. Four isomers of benzene are shown below.

Set up the secular matrix for each of these compounds. You need not extract the secular equation.
The roots of the secular equations and coefficients for the wavefunctions are linked below.
Methylenecyclopentadiene
Trimethylenecyclopropane
1,3-dimethylenecyclobutanediyl
1,2-dimethylenecyclobutene
1. Which of these are expected to be resonance-stabilized? Are any as stable as benzene (based on your computational results)?
2. Which are expected to have significant dipole moments? (Compute atomic charges)
3. Order these in terms of reactivity toward electrophiles. (Look at the HOMOs)
4. Order these interms of reactivity toward nucleophiles. (Look at the LUMOs)
This problem set is due in hard copy by 5 p.m. Friday, Jan. 27.
Back to CH 637 Home page