Coherent diffraction of single molecules embedded in superfluid helium droplets and oriented by a laser field
Rapid progress in the field of genomics and proteomics has pushed the frontier of biological science much closer to the ultimate goal of mechanistic understandings and therapeutic interventions of diseases. Structural information of proteins and protein assemblies is critical because biological activities are essentially governed by their native conformation and conformational flexibility. Currently, of the several types of structural tools available, x-ray crystallography has proven the most prolific for structure determination. However, the difficulty of crystallization poses a significant and sometimes insurmountable obstacle, particularly for protein complexes and membrane proteins.
The conditions for crystallization are highly individualized, and a grand solution should eliminate the need of crystallization altogether. Non-crystalline specimens do not generate discrete Bragg diffraction patterns as single crystals. Instead, they produce speckled continuous Fraunhofer diffraction patterns. Nevertheless, a continuous diffraction pattern should still contain sufficient information for structural determination. Moreover, by sampling the continuous diffraction pattern at a high rate, the phase problem in conventional crystallography can be avoided because of the additional information coded in the known background data. This method is called phase retrieval by oversampling, and it has been not only proven theoretically, but also demonstrated in a few recent experiments.
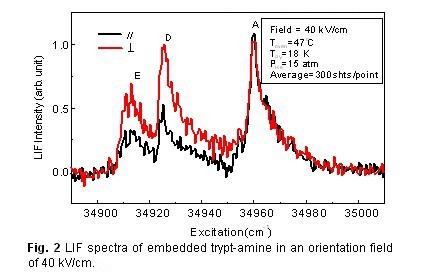
A unique requirement of single molecule imaging is substrate free recording. Our project solves substrate problem by embedding the protein ions in superfluid helium droplets and using a polarized laser for orientation. Figure 1 shows the conceptual design of this project.
.
Several key aspects are crucial to the success of this project: protein ions need to be generated in their native conformation, cooled by superfluid helium droplets, and oriented by a pulsed elliptically polarized laser. The diffraction pattern of a coherent beam of high energy electrons will be sampled at a high frequency for iterative phase retrieval and structural refinement. We have adopted the “divide and conquer” approach, with different subgroups working on different aspects simultaneously. Updates and progresses are available upon request.
Linear dichroism spectroscopy of field oriented molecules
Our laboratory has developed the linear dichroism spectroscopy of oriented gas phase molecules over the past decade. Using a strong DC electric field, polar molecules cooled to temperatures below 1 K either through supersonic expansion or doping in superfluid helium droplets, can be oriented. Polarized excitation of the oriented molecules reveals the direction of the transition dipole or the size and direction of the permanent dipole. For biological molecules that have no resonance lines or too many resonance lines, this type of linear dichroism spectroscopy offers an additional parameter for spectroscopic assignment and tautomeric and conformational identification. This technique enables us to investigate the intrinsic driving force for the formation of secondary structures in peptides.
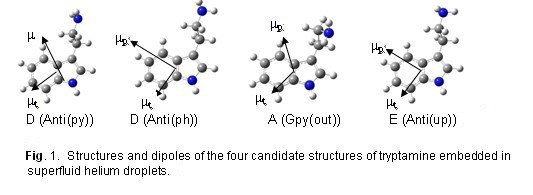
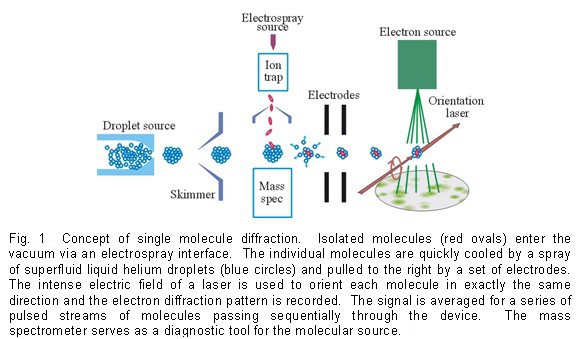
Between the two candidate structures for conformer D, the angle between the two dipoles in the Anti(py) structure is close to the magic angle (180° – 127° = 53°), this angle is close to 90° in the Anti(ph) structure. Our results of linear dichroism spectroscopy revealed strong polarization preference for conformer D, as shown in Figure 2, thus confirming the Anti(ph) structure D. This work exemplifies the power of our experimental approach, since given the similarities of the two candidate structures for conformer D, our approach is the only method currently available for this type of structural determination. Although coupling of the electronic transition with the helium matrix is not negligible, it does not affect the direction of the transition dipole.